Literary Review
There are several journals and research papers that have focused on the effectiveness of 9-aminoacridine and some of its derivatives as cancer treatments. In order to figure out how the 9-aminoacridine works and why it is effective, one must understand how it impacts the DNA on the molecular level. Currently researchers are trying to figure out whether it is a topoisomerase poison or a catalytic inhibitor. A catalytic inhibitor stops the enzyme from working but leaves it there, while a poison just kills the enzyme and gets rid of it all together. Either way, the result is the same, the only difference is how it would be used, or how it will be made to be more effective.
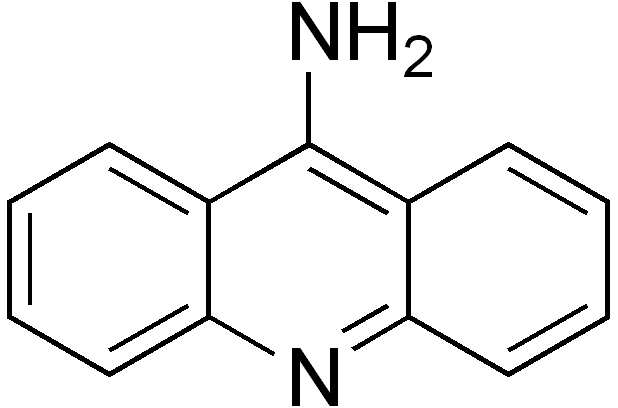
9-aminoacridine:
9-aminoacridine:
Figure 2
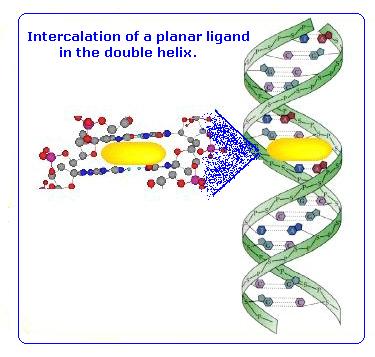
Lots of people have written articles on 9-aminoacridine also known as a DNA intercalator. A DNA intercalator inserts itself between base pairs in the DNA helicase. This means that it can interrupt DNA replication processes. It has also been looked at to see whether it is an antitumor drug. The drug has also been researched and said to be anti-malarial, anti-inflammatory, antimicrobial, and anticancer agents.
DNA Intercalator:
DNA Intercalator:
Figure 3
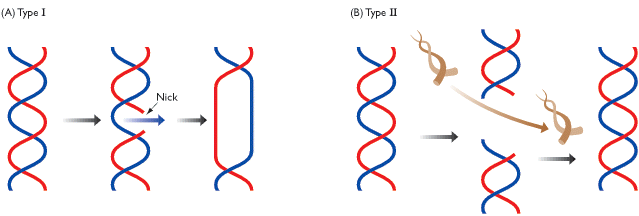
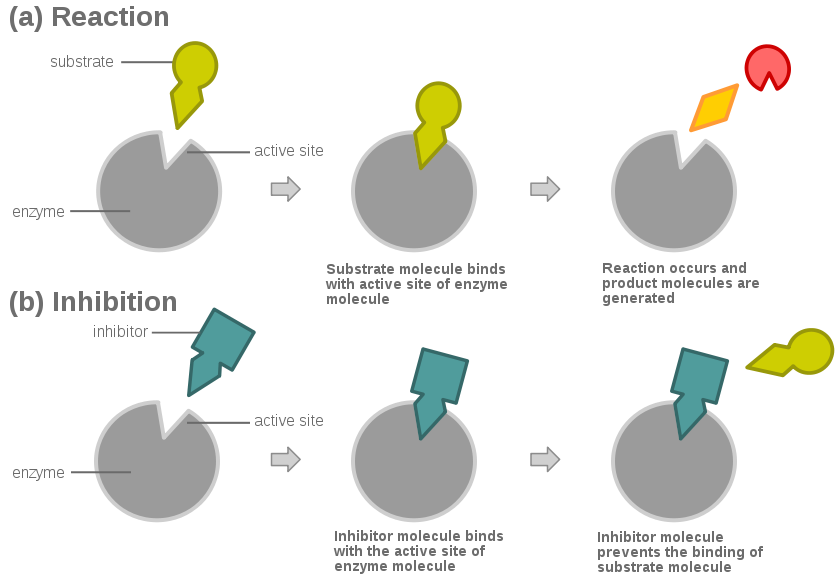
Topoisomerase is an enzyme that alters the way that the DNA is coiled in order to help with the DNA replication process.3 This is essential for the duplication of all cells. Cancer cells replicate at a much faster rate than normal cells so if the topoisomerase is damaged or inhibited, it should make growth of the tumor slow down, and cause apoptosis to begin.
Figure 4
There are 2 different types of topoisomerase. Type one inserts itself into the DNA and then cuts one strand to unwind it were as, type 2 inserts itself and then cuts both of the strands of the DNA and then unwinds it. Lots of research has been done that argues that the 9-aminoacridine acts more like a catalytic inhibitor because it prevents the topoisomerase from working rather than killing the topoisomerase itself. Gálvez-Peralta et al.4 performed an experiment on mice seeing as to how the 9-aminoacridine would work on cells and it seemed as though there was a catalytic inhibitor2 breaking the cancerous cells. However, mice cells are different from human cells, but the DNA structure is similar so the drug should work in the same way for humans. Most of the research that has been done leads to the conclusion that 9-aminoacridine is a catalytic inhibitor.
The catalytic inhibitor might lead to the apoptosis of cells. This is when the cells die because they have been damaged too much. In cancer cells they do not go through apoptosis. Tan et al.7 have found that cancer cells have a gene that prevents them from apoptosis, however, if the DNA cannot replicate, at some point it will go through apoptosis.
The catalytic inhibitor might lead to the apoptosis of cells. This is when the cells die because they have been damaged too much. In cancer cells they do not go through apoptosis. Tan et al.7 have found that cancer cells have a gene that prevents them from apoptosis, however, if the DNA cannot replicate, at some point it will go through apoptosis.
Figure 5
The compound will affect healthy cells and cancer cells in the same way so there is no way to tell for sure that it is only killing the cancer cells. This can lead to many unwanted side effects so it is important to make the drug as effective as possible to make it safer and a more efficient drug compared to chemotherapy. The O-phenyl-N-(9’-acridinyl)-hydroxylamine and its derivatives should be less susceptible to hydrolysis and should be able to kill more cancer with a lower dosage.
Hydrolysis is the breaking up of a molecule using water. Some previously manufactured derivatives of 9-aminoacridine compounds are very susceptible to this because they contain a very weak oxygen carbon bond. The O-phenyl-N-(9’-acridinyl)-hydroxylamine does not have a weak oxygen carbon bond, as compared to carlson’s O-benzyl-N-(9’-acridinyl)-hydroxylamine, is less susceptible to hydrolysis. This is because there is no extra OC bond which is prime spot for hydrolysis to occur. Thus adding a bromine onto the compound, the chances of hydrolysis are not going to change, only the way it attaches to the DNA should change. The bromine substituent is a weak deactivating agent, this will make the entire compound slightly more positive which will make it bond more tighter to the DNA. There are also 3 lone pairs on the bromine, however they will most likely not affect the binding strength. Hydrolysis should also still have some power in the compound because in order to get the drug out of the body in time so then it does not cause too many side effects, it has to break down and not harm the patient’s body any more.
Hydrolysis is when water breaks apart a compound at a weak oxygen to carbon bond. In the compound, there are no weak oxygen to carbon bonds. A similar drug has been made but it was very susceptible to hydrolysis because it had an extra OC bond.
Deselm2 outlined a procedure to make O-phenyl-N-(9’-acridinyl)-hydroxylamine. The method in the methods section is edited for the added bromine substituent.
Hydrolysis is the breaking up of a molecule using water. Some previously manufactured derivatives of 9-aminoacridine compounds are very susceptible to this because they contain a very weak oxygen carbon bond. The O-phenyl-N-(9’-acridinyl)-hydroxylamine does not have a weak oxygen carbon bond, as compared to carlson’s O-benzyl-N-(9’-acridinyl)-hydroxylamine, is less susceptible to hydrolysis. This is because there is no extra OC bond which is prime spot for hydrolysis to occur. Thus adding a bromine onto the compound, the chances of hydrolysis are not going to change, only the way it attaches to the DNA should change. The bromine substituent is a weak deactivating agent, this will make the entire compound slightly more positive which will make it bond more tighter to the DNA. There are also 3 lone pairs on the bromine, however they will most likely not affect the binding strength. Hydrolysis should also still have some power in the compound because in order to get the drug out of the body in time so then it does not cause too many side effects, it has to break down and not harm the patient’s body any more.
Hydrolysis is when water breaks apart a compound at a weak oxygen to carbon bond. In the compound, there are no weak oxygen to carbon bonds. A similar drug has been made but it was very susceptible to hydrolysis because it had an extra OC bond.
Deselm2 outlined a procedure to make O-phenyl-N-(9’-acridinyl)-hydroxylamine. The method in the methods section is edited for the added bromine substituent.
The method given in the methods section is similar to the method that Deselm did except for the added bromine in the benzene and iodobenzene. This should not change the structure of the compound to much, or change its characteristics very much except for how it behaves with the DNA and topoisomerase. The whole thing should be pretty much the same procedure except that the compound will have an added bromine.
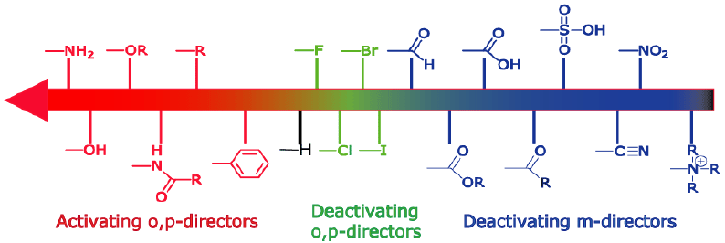
Also, the compound that will be made in this study will be similar to the above except for the added bromine to the benzene and the iodobenzene in the first step. The bromine is a weakly deactivating element but it also has 3 lone pairs. Because of this, it is unsure how the compound will bind with the DNA. This means that it takes away electrons which will make the compound more positively charged, and since DNA has a slight negative charge, it will attach itself better to the DNA if the deactivating part .
Also, the compound that will be made in this study will be similar to the above except for the added bromine to the benzene and the iodobenzene in the first step. The bromine is a weakly deactivating element but it also has 3 lone pairs. Because of this, it is unsure how the compound will bind with the DNA. This means that it takes away electrons which will make the compound more positively charged, and since DNA has a slight negative charge, it will attach itself better to the DNA if the deactivating part .
Figure 7
Deactivating means that the substituent group shifts the center of electronegativity.
The first 2 steps to the procedure involve the synthesis of N-aryloxyimides and aryloxamines. These steps have been outlined by Ghosh et al5 and Deselm. Ghosh et al talked about methods to figure out what the compound is by using a H-NMR. The procedure done by Deselm uses iodobenzene and benzene, in the procedure, a derivative will be used. Iodobenzene and nitrobenzene will be used in order to figure out how it will affect the efficacy of the end product.
The product of step 3 that is needed to perform step 4 can be bought from pharmaceutical companies, however, manufacturing that compound in the lab is more cost effective. In order to make this drug more affordable for people to use/purchase, it needs to be made in the most cheap way possible which also gives the most amount of concentration of the desired product.
There are several ways to check if the compound was made correctly and then to see if there is enough of it in the solution. These methods include using temperature gradient gel electrophoresis (TGGE), IR, and 1H-NMR. While using the TGGE it will also find out how the drug will interact with the DNA and how it will affect tumor cells.
The 9-aminoacridine is very important and is proving itself to be effective for several different types of cancer. The currently existing versions of this compound are very susceptible to hydrolysis which is why a derivative should be better in that aspect. Since the main thing that needs to be improved is this, there is a need to make the O-phenyl-N-(9’-acridinyl)-hydroxylamine and its derivatives as proposed.
The first 2 steps to the procedure involve the synthesis of N-aryloxyimides and aryloxamines. These steps have been outlined by Ghosh et al5 and Deselm. Ghosh et al talked about methods to figure out what the compound is by using a H-NMR. The procedure done by Deselm uses iodobenzene and benzene, in the procedure, a derivative will be used. Iodobenzene and nitrobenzene will be used in order to figure out how it will affect the efficacy of the end product.
The product of step 3 that is needed to perform step 4 can be bought from pharmaceutical companies, however, manufacturing that compound in the lab is more cost effective. In order to make this drug more affordable for people to use/purchase, it needs to be made in the most cheap way possible which also gives the most amount of concentration of the desired product.
There are several ways to check if the compound was made correctly and then to see if there is enough of it in the solution. These methods include using temperature gradient gel electrophoresis (TGGE), IR, and 1H-NMR. While using the TGGE it will also find out how the drug will interact with the DNA and how it will affect tumor cells.
The 9-aminoacridine is very important and is proving itself to be effective for several different types of cancer. The currently existing versions of this compound are very susceptible to hydrolysis which is why a derivative should be better in that aspect. Since the main thing that needs to be improved is this, there is a need to make the O-phenyl-N-(9’-acridinyl)-hydroxylamine and its derivatives as proposed.